
Help test an investigational drug to see if it can safely block the immune response to gluten that causes harm in people with a celiac disease.
Is celiac disease holding you back? If so, consider joining the FB102-301 study for people who have celiac disease who are currently on a gluten-free diet. The phase 2 FB102-301 study is researching a potential new drug for celiac disease.
Who can join
the study?
You may be able to join the study if you meet these criteria:
- 18 to 70 years old
- Diagnosed with celiac disease (confirmed by biopsy and blood test at least 1 year ago)
- Have been on a gluten-free diet for at least
1 year and are willing to continue - Not actively experiencing signs or symptoms of celiac disease
What do participants receive?
- Possible access to the study drug
- 1 in 5 participants receives a placebo, which looks like the study drug but does not have active ingredients
- The placebo group is important for comparing results at the end of the study
- The opportunity to contribute to research on celiac disease
- Reimbursement for meals and travel
- Compensation for time and travel
What is
the study?
The FB102-301 study is testing an investigational study drug called FB102.
The goal of the study is to find out whether FB102 can safely reduce the harmful effects of gluten in adults with celiac disease.
Investigational means FB102 has not been approved by any health or regulatory agencies. It can only be used in clinical research studies.
More about study drug FB102
FB102 is a type of drug called a monoclonal antibody. It is designed to block signals the immune system sends when it detects gluten. Blocking these signals may prevent gluten from causing inflammation and damage in the small intestine.
What are clinical research studies?
Clinical research studies, like the FB102-301 study, help scientists and doctors explore whether a drug is safe and whether it works. Before a doctor can prescribe a new drug, it must go through several phases of clinical research:
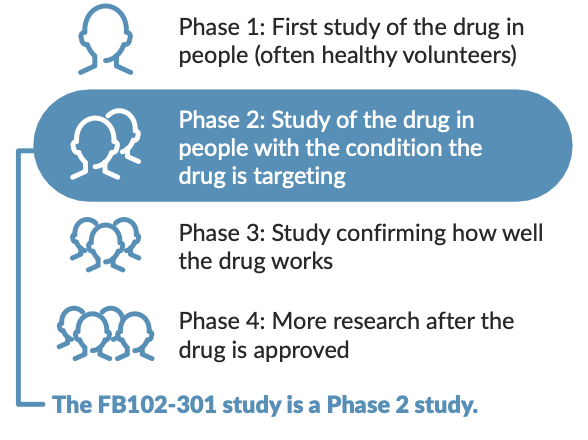
What can participants expect?

Screening
- You will have study assessments to find out if you qualify.
- Study assessments may include, but are not limited to, a physical exam (with height, weight, and vital signs), a heart test (ECG), blood tests, an esophagogastroduodenoscopy (EGD) with biopsies, and questions about your health and celiac disease symptoms.
- For an EGD, you will receive sedation or anesthesia to make you relax or sleep during the procedure.
Study Drug
- You will receive intravenous (IV) infusions of the study drug or placebo into a vein in your arm or hand.
- For the first 5 weeks, you will have 1 infusion every week at the study clinic.
- For the last 6 weeks, you will have 1 infusion every 2 weeks at the study clinic.
- From Weeks 3 to 11, your study doctor will guide you through a gluten challenge. By reintroducing small
- amounts of gluten, the study doctor will be able to tell if the infusions work as expected.
- You will have regular visits throughout this period for study assessments, including a final EGD with biopsy.
Follow-Up
- You will have 2 study visits for study assessments.
- The study doctor may ask if you would like to take part in a separate, optional long-term follow-up study.
What is the study drug?
The investigational study drug, FB102, is a monoclonal antibody. It is designed to block signals the immune system sends when it detects gluten. Blocking these signals may prevent inflammation and damage in the small intestine caused by gluten ingestion in people with celiac disease.
Will I receive the study drug?
Approximately 4 out of every 5 participants (80%) will receive the study drug. The other 20% of participants will receive placebo, which looks like and is administered the same as the study drug but does not have active ingredients. The placebo group is important for comparing results at the end of the study.
Pre-qualify today
Join the FB102-301 study and help progress research for a potential new celiac disease treatment.
