Compensation for you time and travel
Is your blood pressure medication not working?
If your blood pressure is high despite blood pressure medication, find out if you qualify for the HALO study.
The goal of the HALO study is to find out if a study drug can safely lower blood pressure in
adults with uncontrolled hypertension (high blood pressure). Uncontrolled means a person’s
blood pressure is high, even though they are taking a medication for it.
What is the study drug?
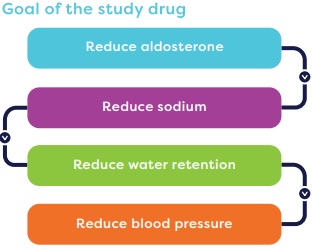
The study drug is designed to reduce aldosterone levels. Reducing aldosterone may reduce the amount of sodium and water in the blood and lead to a lower blood pressure.
The study drug is investigational, which means that it is not approved by the US Food and Drug Administration (FDA) and can only be used in clinical research studies.
Goal of the study drug
- Reduce aldosterone
- Reduce sodium
- Reduce water retention
- Reduce blood pressure
More about aldosterone
Aldosterone helps balance how much sodium and potassium are in the blood. When the body produces too much aldosterone, it can cause the blood to hold more sodium than it should. Because sodium binds to water, extra sodium in the blood means there will be extra water in the blood, too. More fluid inside the veins increases the pressure on the veins. This is known as high blood pressure.
Who can join the study?
You may be able to join the HALO study if you meet the following study requirements:
• You are 18 years old or older with high blood pressure.*
• You already take 1 medication for high blood pressure.
• You may have high levels of aldosterone in your blood.*
*Your blood pressure and aldosterone levels will be measured as part of this study to find out if you qualify.
What can study participants expect?
The HALO study lasts about 18 weeks (about 4 and a half months), and you can expect the following:
- Screening (up to 2 weeks): Visit the study clinic to have study assessments to find out if you qualify.
- Run-in (up to 4 weeks): Take a placebo tablet (no active ingredients) once a day in addition to your regular blood pressure medication.
- Dosing Part 1 (8 weeks): Be randomly assigned to take study drug or placebo tablets daily in addition to your regular blood pressure medication. Visit the study clinic 5 times for study assessments. If you meet certain requirements, you may be asked to participate in the next period of the study (Dosing Part 2). If not, you will skip to the Follow-up period.
- Dosing Part 2 (4 weeks): Continue taking the study drug (possibly at a higher dose) or placebo and stop taking your regular blood pressure medication. Visit the study clinic 3 times for study assessments. You may be asked to join a separate, related study and skip the Follow-up period of this study.
- Follow-up (2 weeks): Visit the study clinic 1 more time for final study assessments.
Eligible study participants receive at no cost:
- Possible access to a study drug.
- Some participants will receive a placebo, which has no active ingredients.
- Participants who receive a placebo during the first part of the study may be able to take the study drug in the second part if they meet certain criteria.
- Current blood pressure medication (paid for by the study.
- Possible compensation for time and travel.
- Study support and monitoring by a healthcare team.
- Information about high blood pressure.
If you want to know more about it, like What can study participants expect?, you can download the study brochure.
